About the Project
The project involved gathering requirements from various departments to ensure the portal met diverse needs. We designed a user-friendly interface that included features such as document libraries, discussion forums, and event calendars. Training sessions were conducted to help employees adapt to the new system. Post-implementation surveys indicated a significant improvement in employee satisfaction and collaboration.
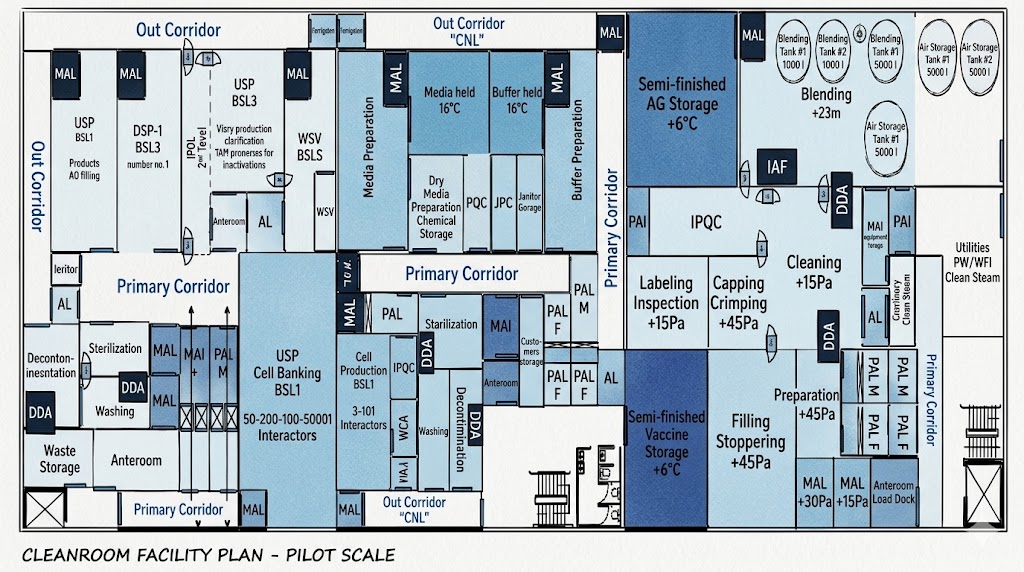
Mixed Viral/Bacterial Facility: CAPA & Risk Assessment for Shared Areas
Shared filling area CAPA + risk assessment package
Mixed Viral/Bacterial Facility: CAPA & Risk Assessment for Shared Areas
Mixed viral/bacterial facility, shared filling area

What We Did
1
Built CCS-aligned risk assessment (segregation, campaign planning, controls)
1
Developed CAPA table with clear owners, evidence, timelines
1
Prepared audit response narrative emphasizing practical risk controls and verification

Deliverables
1
One-page audit response letter (professional, evidence-oriented)
1
CAPA table (controls, verification, responsibilities, deadlines)
1
Mini risk assessment outline (QRM logic, hazards, mitigations, residual risk)
Outcome
Stronger compliance posture, reduced audit risk, improved operational clarity.