Typical Outcomes
Receive accurate diagnoses that guide treatment decisions.
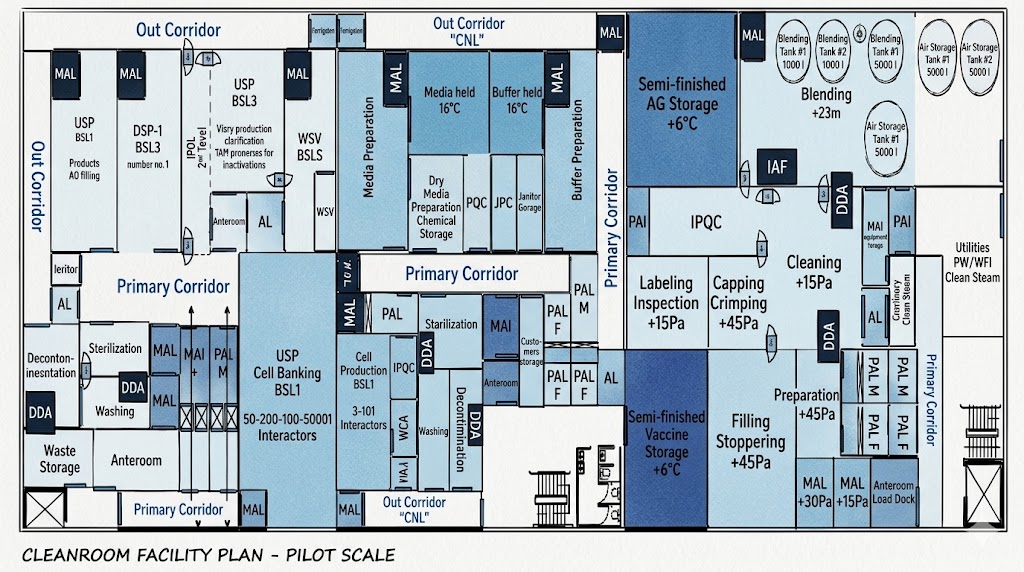
Defensible Segregation & Contamination Control
Robust strategy for cleanroom zoning and cross-contamination prevention
URS That Vendors Can Quote Accurately
Comprehensive User Requirement Specifications for precise vendor responses
Commissioning/Qualification Readiness
Structured approach to FAT/SAT and validation activities
Deliverables
Detailed pathology reports will be provided for your review.
Draft Design Audit
Layout, flows, zoning, segregation analysis
Cleanroom/HVAC Concept Inputs
Pressures, classifications, airflow requirements
Utilities Concept Inputs
PW/WFI, clean steam, gases, waste systems
Equipment URS Packages
Bioreactors, harvest, inactivation, formulation, filtration, filling, lyophilization, washers, autoclaves
FAT/SAT Test Plans
Factory & Site Acceptance Testing with acceptance criteria
Validation Readiness Outline
VMP structure, IQ/OQ approach and protocols
CCS-Aligned Risk Assessment
Cross-contamination and mix-up prevention strategies
Platforms We Support
Available through various healthcare platforms.
mRNA / LNP
Advisory support
Used in:

Human

Vet

Typical Risks / Bottlenecks
- Supply chain (lipids, enzymes)
- Complex analytics (size, encapsulation, integrity)
- Aseptic manufacturing strategy
- Cold chain logistics & stability

What We Do
- CMC roadmap & regulatory strategy
- Facility concept (isolators, aseptic)
- Vendor shortlist (lipids, CDMO)
- QA/QC readiness & analytics build

Typical Deliverables

Target Product Profile / CMC Outline

Feasibility Report
Baculovirus / VLP
Virus-like particle platforms
Used in:

Human

Vet

Typical Risks / Bottlenecks
- Lot-to-lot consistency
- Complex impurity profile (baculovirus, host proteins)
- Scale-up challenges (cell density, MOI)
- Analytical method development (empty vs full VLP)

What We Do
- Upstream strategy (insect cell platform, MOI optimization)
- DSP train design (clarification, chromatography, TFF)
- Analytical method map (EM, SEC, potency)
- Tech transfer from R&D to pilot/commercial

Typical Deliverables

Upstream process optimization plan

DSP flowsheet & unit operation selection

Analytical control strategy

Tech transfer package & training
Industries
Serving hospitals, clinics, and individual patients.
Human Vaccines

Typical Projects
- Supply chain (lipids, enzymes)
- Complex analytics (size, encapsulation, integrity)
- Aseptic manufacturing strategy
- Cold chain logistics & stability

What Success Looks Like
- Expression platform comparison & selection
- Upstream & downstream integration
- Formulation & adjuvant compatibility
- Stability & aggregation risk mitigation

Common Pitfalls
- Supply chain (lipids, enzymes)
- Complex analytics (size, encapsulation, integrity)
- Aseptic manufacturing strategy
- Cold chain logistics & stability

Cost-of-goods

Cost-of-goods

Cost-of-goods

Cost-of-goods
Human Vaccines
Veterinary Vaccines

Typical Projects
- Supply chain (lipids, enzymes)
- Complex analytics (size, encapsulation, integrity)
- Aseptic manufacturing strategy
- Cold chain logistics & stability

What Success Looks Like
- Expression platform comparison & selection
- Upstream & downstream integration
- Formulation & adjuvant compatibility
- Stability & aggregation risk mitigation

Common Pitfalls
- Supply chain (lipids, enzymes)
- Complex analytics (size, encapsulation, integrity)
- Aseptic manufacturing strategy
- Cold chain logistics & stability

Cost-of-goods

Cost-of-goods

Cost-of-goods

Cost-of-goods
Veterinary Vaccines
Insights & Resources
Industry expertise, best practices, and thought leadership in vaccine development
Add-on Services
Follow-up consultations available for further discussion.
Training (Production / QC / QA / Engineering)
Tailored workshops with templates, quizzes, and 'audit-friendly' outputs
Deliverables
- Gap report
- CAPA register
- Risk assessment outline
- Evidence package
What we need from you
Tissue samples and relevant medical history.
Gap assessment / Risk-Based Roadmap (QRM/CCS)
Risk mapping and prioritization
- Identify critical risks (contamination, mix-ups, segregation gaps, utilities, validation readiness)
- Turn risks into a practical plan
Let's discuss your vaccine project with
Lab to Jab
Tell us what you're planning, building, and where you are today. We'll respond with the fastest path to a defensible plan.
Thank you! Your submission has been received!
Oops! Something went wrong while submitting the form.